The integration of metabolomics into clinical trials is revolutionizing medical research by providing detailed insights into the biochemical changes associated with various diseases and treatments. Metabolomics, the comprehensive study of small molecules (metabolites) in biological samples, is increasingly being used to understand disease mechanisms, identify biomarkers, and evaluate therapeutic responses.
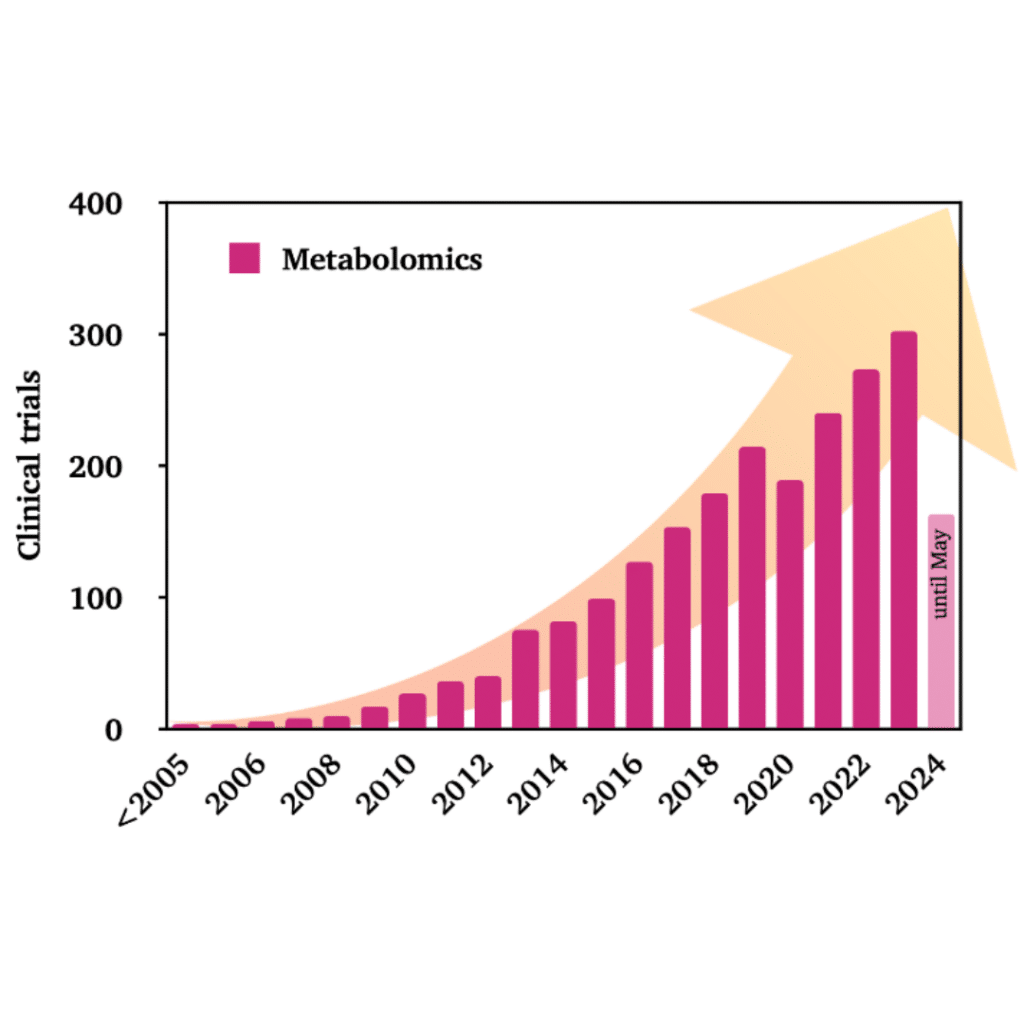
A search in the most used database for clinical trials (ClinicalTrials.gov) with the term “Metabolomics” returns more than 2,000 studies. Since the first studies were registered before 2005, every year has seen more studies documented than the previous one. The only exception being 2020 due to the COVID-19 pandemic.
In 2023, more than 300 clinical trials were posted, and 2024 looks promising as well, with more than 160 studies filed by May, compared to 123 studies during the same period in 2023. In fact, among all 2024 studies from all fields, nearly 1% already include metabolomics as part of the study.
With around 900 clinical studies completed in the last 15 years, 835 investigations are currently seeking participants, split roughly equally between observational and interventional studies. Among the latter, according to the stages defined by the FDA, 15 clinical trials are categorized as phase 4, 14 as phase 3, 57 as phase 2, 23 as phase 1, and 10 as early phase 1.
Regarding the funding type, 42 clinical trials are funded by the industry, such as pharmaceutical and device companies, 49 by various U.S. Federal Agencies, and 423 by universities and organizations.
Metabolomics in Clinical Trials
Metabolomics is instrumental in identifying biomarkers that can predict disease risk, progression, and response to therapy. By understanding the unique metabolic profiles of individuals, metabolomics enables the development of personalized treatment strategies. The dynamic nature of metabolomic profiles allows for real-time monitoring of treatment efficacy and safety by providing insights into the mechanisms underlying disease and treatment effects.
Accuracy of risk evaluation and treatment approaches:
With the use of NMR, physicians can obtain a clearer and more complete picture of the patients’ lipid profile, which aids in designing more precise and appropriate interventions that address not only the symptoms of lupus but also the associated long-term risks, such as cardiovascular diseases. This type of advanced diagnosis is a step towards personalized medicine, where treatment and prevention are specifically tailored to the individual characteristics of the patient, enhancing the effectiveness of interventions and minimizing adverse effects.
In summary, NMR metabolomics establishes itself as a fundamental pillar in the fight against the secondary complications of lupus, opening new paths for the management and treatment of this complex disease.
Case Studies
Several clinical trials have successfully integrated metabolomics to enhance their findings:
- Cardiovascular Disease. Metabolomic studies have identified key metabolic changes associated with the progression of cardiovascular diseases. For instance, targeted lipid metabolomics has been used to assess the impact of specific treatments on lipid metabolism and cardiovascular risk (NCT03837925, NCT04551872).
- Cancer. Metabolomics has been used to identify metabolic biomarkers for early cancer detection and to monitor the effectiveness of chemotherapy and other treatments. This helps in tailoring treatments to individual metabolic responses (NCT03736993, NCT01111097).
- Obesity and Bariatric Surgery. Research has shown that metabolomic profiling can elucidate the metabolic changes following bariatric surgery. It highlights improvements in lipid profiles and reductions in cardiovascular risk factors (NCT03181347, NCT02671279).
Future Perspectives
The future of metabolomics in clinical trials looks promising, with several advancements on the horizon:
- Enhanced Analytical Techniques. Continuous improvements in NMR and MS technologies will increase the accuracy and throughput of metabolomic analyses.
- Integration with Other Omics. Combining metabolomics with genomics, proteomics, and transcriptomics will provide a more comprehensive understanding of biological systems.
- Artificial Intelligence. Machine learning algorithms will play a crucial role in analyzing complex metabolomic data, identifying patterns, and making predictive models.
In conclusion, metabolomics is set to become an integral part of clinical trials. It offers a powerful tool for understanding disease mechanisms, developing personalized therapies, and improving patient outcomes. As this field continues to evolve, it holds the potential to transform medical research and clinical practice, paving the way for more effective and targeted healthcare solutions.
